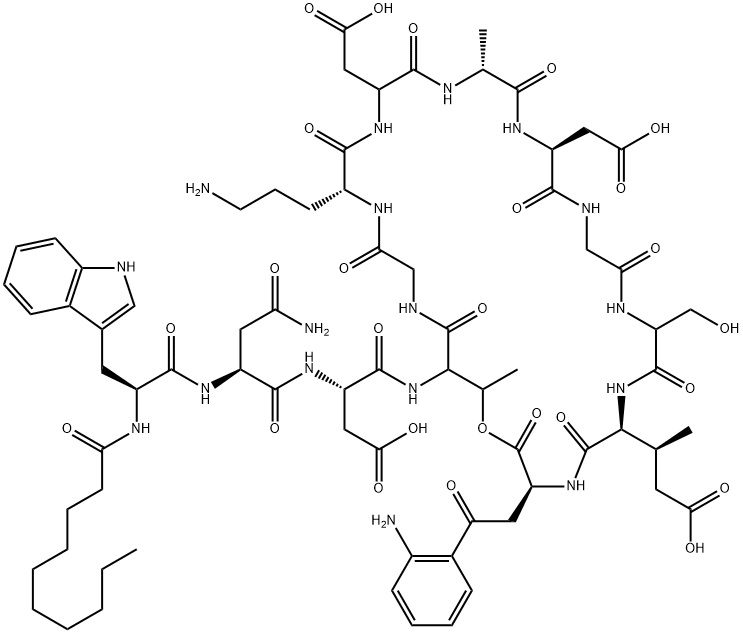
Daptomycin , ≥98% , 103060-53-3
Synonym(s):
Dapcin;N-decanoyl-L-tryptophyl-L-asparaginyl-L-aspartyl-L-threonylglycyl-L-ornithyl-L-aspartyl-D-alanyl-L-aspartylglycyl-D-seryl-threo-3-methyl-L-glutamyl-3-anthraniloyl-L-alanine[egr]1-lactone
CAS NO.:103060-53-3
Empirical Formula: C72H101N17O26
Molecular Weight: 1620.67
MDL number: MFCD08282794
EINECS: 600-389-2
| Pack Size | Price | Stock | Quantity |
| 10MG | RMB87.20 | In Stock |
|
| 50MG | RMB319.20 | In Stock |
|
| 100MG | RMB375.20 | In Stock |
|
| 250mg | RMB559.20 | In Stock |
|
| 1G | RMB1409.60 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 202-204?C |
| Boiling point: | 2078.2±65.0 °C(Predicted) |
| Density | 1.45±0.1 g/cm3(Predicted) |
| Flash point: | 87℃ |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | methanol: soluble5mg/mL |
| pka | 4.00±0.10(Predicted) |
| form | powder |
| color | colorless to faint yellow |
| biological source | Streptomyces roseosporus |
| λmax | 260nm(EtOH)(lit.) |
| Merck | 14,2823 |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. |
| InChIKey | DOAKLVKFURWEDJ-RWDRXURGSA-N |
Description and Uses
Daptomycin is the first entry of a new class of cyclic lipopeptide antibiotics that disrupts multiple aspects of bacterial membrane function including disruption of membrane potential and amino acid transport, inhibition of lipoteichoic acid synthesis and inhibition of peptidoglycan synthesis. It is indicated for the treatment of complicated skin and skinstructure infections (cSSSI) caused by a range of Gram-positive bacteria. This is distinct from previous classes of antibiotics that inhibit bacterial cell wall biosynthesis, bacterial DNA replication, and folate coenzyme biosynthesis. Due to this unique mechanism, cross-resistance has not been noted with any other class of antibiotics. It is produced by the fermentation of Streptomyces roseporus. The fatty acid side chain is a key determinant of acute toxicity, with the ten-carbon chain least acutely toxic to mice. Daptomycin has shown efficacy in a variety of animal models versus several Grampositive infectious agents including methicillin-susceptible S aureus, vancomycinresistant E faecalis, spyogenes and S pneumoniae. It retains in vitro activity against methicillin, vancomycin and linezolid-resistant strains including Staphylococcus aureus (MRSA and VRSA), which is the leading cause of hospital-acquired infections (nosocomial infections (Nis)). The MIC values against Gram-positive pathogens are relatively low, ranging from 0.06 to 2.0 mg/mL. In two clinical studies treating patients (ca. 1090 for both arms of both studies) with complicated skin and soft tissue infections (cSSTIs) in which gram-positive pathogens were suspected and parenteral antibiotics were required, daptomycin provided similar clinical success rates as compared to standard therapy with vancomycin or semisynthetic penicillins such as cloxacillin, oxacillin, or flucloxacillin. The daptomycin treated group showed more rapid improvement as noted by scoring on day three or four and also had a shorter duration of treatment versus the standard therapy group (7 vs 8 days). It is dosed once daily (4 mg/kg/day) by intravenous infusion and has a half-life of 8.1 h. It is primarily cleared renally and thus requires dosing adjustments for those with severe renal insufficiency (CLCR ,30 mL/min). Clinical safety of daptomycin is similar to other antibiotics.
immunosuppressant
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P280-P302+P352-P362+P364 |
| WGK Germany | 3 |
| RTECS | HB5626000 |
| HS Code | 29419090 |
| Toxicity | LD50 i.v. in mice: 600 mg/kg (Debono) |