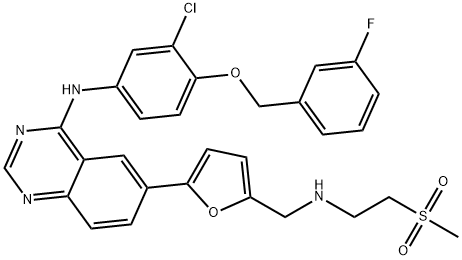
Lapatinib , ≥99% , 231277-92-2
Synonym(s):
;Lapatinib;N-(3-Chloro-4-((3-fluorophenyl)methoxy)phenyl)-6-(5-(((2-(methylsulfonyl)ethyl)amino)methyl)-2-furanyl)-4-quinazolinamine;N-(3-Chloro-4-((3-fluorophenyl)methoxy)phenyl)-6-(5-((2-methylsulfonylethylamino)methyl)-2-furyl)quinazolin-4-amine
CAS NO.:231277-92-2
Empirical Formula: C29H26ClFN4O4S
Molecular Weight: 581.06
MDL number: MFCD09264194
EINECS: 1806241-263-5
| Pack Size | Price | Stock | Quantity |
| 25MG | RMB55.20 | In Stock |
|
| 100MG | RMB104.80 | In Stock |
|
| 250MG | RMB157.60 | In Stock |
|
| 500MG | RMB167.20 | In Stock |
|
| 1G | RMB291.20 | In Stock |
|
| 5G | RMB1439.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 144-146oC |
| Boiling point: | 750.7±60.0 °C(Predicted) |
| Density | 1.381±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C(protect from light) |
| solubility | Soluble in DMSO (up to 200 mg/ml) |
| form | solid |
| pka | 6.34±0.19(Predicted) |
| color | Yellow |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 1 month. |
| InChIKey | BCFGMOOMADDAQU-UHFFFAOYSA-N |
| SMILES | N1=C2C(C=C(C3=CC=C(CNCCS(C)(=O)=O)O3)C=C2)=C(NC2=CC=C(OCC3=CC=CC(F)=C3)C(Cl)=C2)N=C1 |
| CAS DataBase Reference | 231277-92-2(CAS DataBase Reference) |
Description and Uses
Lapatinib, a new member of the 4-anilinoquinazoline class of RTK inhibitors
(RTKIs), was launched as an oral treatment for breast cancer.
Lapatinib has dual affinity for EGFR and HER2 tyrosine kinases. It is indicated
in combination with capecitabine for treating patients with advanced or
metastatic breast cancer whose tumors overexpress HER2 and who have
received prior therapy including an anthracycline, a taxane, and trastuzumab.
Previously marketed drugs from the 4-anilinoquinazoline class include
erlotinib (Tarceva) and gefitinib (IressaTM), both of which are indicated for
treating non-small-cell lung cancer (NSCLC). As with erlotinib and gefitinib, To Market, To Market 2007 475 lapatinib is an ATP-competitive kinase inhibitor. It inhibits the tyrosine kinase activity EGFR and HER-2 with apparent Ki values of 3 and 13 nM, respectively, and has slow off-rate kinetics (t1/2X300 min).
In addition, dividing the daily dose of lapatinib results in approximately 2-fold higher exposure at steady state compared to the same total dose administered once daily.
The chemical synthesis of lapatinib entails the condensation of 4-chloro-6-iodoquinazoline and 3-chloro-4-(3-fluorobenzyloxy)aniline to produce a diaryl amine intermediate followed by Stille coupling of the iodo group with 5-dioxolanyl-2-(tributylstannyl)furan and subsequent acid hydrolysis of the cyclic ketal to the corresponding aldehyde. Finally, reductive amination of the aldehyde intermediate with 2-(methanesulfonyl) ethylamine in the presence of sodium triacetoxyborohydride produces lapatinib.
.
Lapatinib, used in the form of Lapatinib Ditosylate, is a potent EGFR and ErbB2 inhibitor with IC50 of 10.8 and 9.2 nM, respectively. It is a antineoplastic which is used in breast cancer research and a tyrosine kinase inhibitor. It is used for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 (ErbB2)
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H319-H362-H413 |
| Precautionary statements | P201-P260-P263-P273-P305+P351+P338-P308+P313 |
| HS Code | 29349990 |
| Hazardous Substances Data | 231277-92-2(Hazardous Substances Data) |