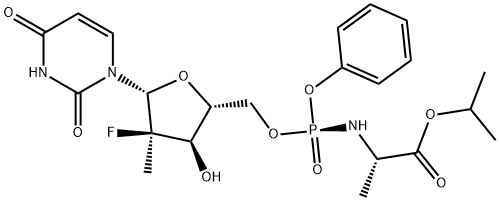
Etoricoxib , ≥98.0% , 202409-33-4
Synonym(s):
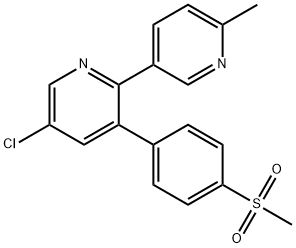
5-Chloro-3-[4-(methylsulfonyl)phenyl]-2-(2-methyl-5-pyridinyl)pyridine;5-Chloro-6′-methyl-3-[4-(methylsulfonyl)phenyl]-2,3′-bipyridine
CAS NO.:202409-33-4
Empirical Formula: C18H15ClN2O2S
Molecular Weight: 358.84
MDL number: MFCD06797512
EINECS: 682-421-5
| Pack Size | Price | Stock | Quantity |
| 10MG | RMB23.20 | In Stock |
|
| 50MG | RMB47.20 | In Stock |
|
| 250MG | RMB72.80 | In Stock |
|
| 1g | RMB230.40 | In Stock |
|
| 5g | RMB783.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 134-135°C |
| Boiling point: | 510.0±50.0 °C(Predicted) |
| Density | 1.298±0.06 g/cm3(Predicted) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | DMSO : 100 mg/mL (278.68 mM; Need ultrasonic) |
| pka | 4.5(at 25℃) |
| form | Solid |
| color | Light yellow to yellow |
| BRN | 8073797 |
| InChIKey | MNJVRJDLRVPLFE-UHFFFAOYSA-N |
| CAS DataBase Reference | 202409-33-4(CAS DataBase Reference) |
Description and Uses
Etoricoxib is a COX-2 inhibitor developed as a follow-up of rofecoxib for the treatment of osteoarthritis, rheumatoid arthritis, dysmenorrhoea, gout, ankylosing spondylitis and pain. Several processes describe the preparation of etoricoxib in 4 or 5 steps from 6- methylnicotinate. The key step is the novel pyridine construction using annulation of a ketosutfone with a vinamidinium synthon. In human whole blood, in vitro, the IC50 value obtained for inhibition of COX-2 is 1 .I μM as compared to 116 μM obtained for inhibition of COX-1. Thus, etoricoxib is the most selective COX-2 inhibitor to date, with a COX-IKOX- 2 ratio of IC50 values of 106 for etoricoxib as compared to 35, 30, 7.6 for rofecoxib, valdecoxib and celecoxib, respectively. Its in vivo potency is generally comparable to that of rofecoxib in animal models against inflammation (carrageenan-induced paw edema), pyrexia (LPS-induced pyresis), pain (carrageenan-induced hyperalgesia) and arthritis (adjuvant-induced arthritis). Etoricoxib is well tolerated with dose-proportional pharmacokinetics. It has no effect on bleeding time or platelet ag regation. The gastrointestinal tolerability of etoricoxib is excellent as demonstrated by [51Cr] models of excretion in rats and squirrel monkeys. Moreover, etoricoxib, unlike naproxen is not associated with significant inhibition of gastric mucosal PGE2 synthesis compared to placebo. Etoricoxib is highly absorbed, has a tmax of 1.5 h and a half-life time of approximately 15-22h. Five metabolites, weak inhibitors of COX-1 and COX-2 have been identified after renal excretion. Finally, although multiple CYP enzymes are involved in the metabolism of etoricoxib (CYP3A4 being the major contributor), etoricoxib is not a potent CYP3A4 inhibitor or inducer. In patients undergoing molar extraction, etoricoxib showed similar efficacy to naproxen sodium with a longer duration of analgesia than acetaminophen/codeine (approximately >24 h, 22 h and 5.2 h, respectively) and a better total pain relief score over 8 h. Similar efficacy of etoricoxib and naproxen was also seen in patients suffering of osteoarthritis. In the treatment of rheumatoid arthritis and ankylosing spondylitis, etoricoxib demonstrated significantly superior efficacy compared to naproxen and placebo. Etoricoxib did not affect the pharmacokinetics of prednisolone (i.v. or p.0.) and its co-administration with antacids showed insignificant effects on the maximal concentration and its absorption. .
For the treatment of rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, chronic low back pain, acute pain and gout.