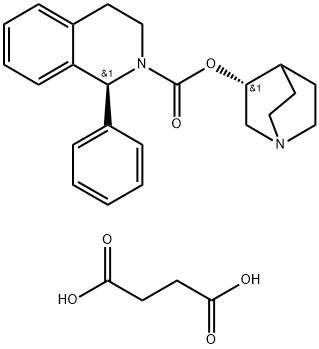
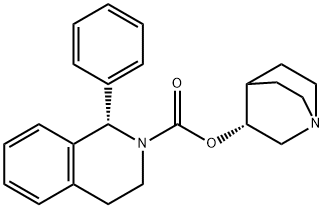
Solifenacin Succinate , >98.0%(HPLC) , 242478-38-2
CAS NO.:242478-38-2
Empirical Formula: C27H32N2O6
Molecular Weight: 480.56
MDL number: MFCD09952287
EINECS: 620-505-5
| Pack Size | Price | Stock | Quantity |
| 25mg | RMB188.00 | In Stock |
|
| 100mg | RMB612.00 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | ~145° |
| storage temp. | under inert gas (nitrogen or Argon) at 2-8°C |
| solubility | Methanol (Slightly, Heated, Sonicated), Water (Slightly, Sonicated) |
| form | Solid |
| color | White to Off-White |
| optical activity | [α]/D +35 to +45°, c =1 in H2O |
| Water Solubility | H2O: 2mg/mL, clear |
| Merck | 14,8712 |
| Stability: | Hygroscopic |
| CAS DataBase Reference | 242478-38-2(CAS DataBase Reference) |
Description and Uses
Solifenacin is an M3 muscarinic receptor antagonist that was developed and launched for the treatment of overactive bladder (pollakiuria) in Europe. M3 receptors have been implicated in neurally evoked smooth muscle contractions of the bladder, and M2 receptors have also been suspected of playing a role because of their dominance in the detrusor muscle. Solifenacin displays affinity for both M3 and M2 receptors with Ki values of 9.9nM and 120 nM, respectively. Since muscarinic salivary glands are of the M3 persuasion, a common side effect of antimuscarinic therapy is dry mouth. At the cellular level, solifenacin possesses a selective preference for bladder over salivary gland that is 15-fold greater than that of atropine suggesting a lower probability of inducing dry mouth at pharmacologically relevant doses. The synthesis of solifenacin involves the preparation of racemic 1-phenyl- 1,2,3,4-tetrahydroisoquinoline via cyclization of N-(2-phenylethyl)benzamide, and subsequent reaction with ethyl chloroformate and transesterification with (R)- 3-quinuclidinol. Chiral chromatography affords the isolation of the desired diastereomer. Alternatively, 1-phenyl-1,2,3,4-tetrahydroisoquinoline may be subjected to optical resolution with (+)-tartaric acid prior to treatment with ethyl chloroformate and subsequent transesterification. The pooled results of four phase III trials concluded that 63% of women receiving 5mg of solifenacin once daily and 68% of women receiving 10mg once daily reported a 50% or more reduction in urgency episodes, compared to 44% of women taking placebo. This compares with a 53% reduction in patients receiving tolterodine twice daily. In another placebo-controlled trial, with the change in the number of micturitions in a 24-h period as the primary endpoint, once-daily solifenacin recorded an 18% decrease for a 5-mg dose and a 21% decrease for a 10-mg dose compared to 10% with placebo. Pharmacokinetic studies have demonstrated that solifenacin has an oral bioavailability of 90%, a long elimination half-life (50 h), low clearance (9.39 L/h), a mean Vss of 599 L, a Cmax of approximately 14 ng/mL, and a time to maximal plasma concentration of 4 h making it suitable for q.d. dosing. Furthermore, these PK parameters are not affected by food ingestion. Solifenacin is excreted predominantly in the feces with only 3–6% found in urine. It is contraindicated in patients with hepatic impairment, gastric retention, urinary retention, or uncontrolled narrow angle glaucoma. Further precautions, such as dose adjustment, should be considered for patients with concurrent use of ketoconazole or other potent CYP3A4 inhibitors or for patients with a history of QT prolongation or currently on medications known to prolong the QT interval. Finally, while other muscarinic antagonists have been explored in the treatment of irritable bowel syndrome (IBS), it is too early to predict the therapeutic utility of solifenacin for IBS although animal studies are promising.
sedative
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P501-P270-P264-P301+P312+P330 |
| RTECS | NW7136000 |
| HS Code | 29339900 |