PRODUCT Properties
| Melting point: | 152-153° |
| alpha | D20 -53° (in 95% ethanol) |
| Boiling point: | 450.59°C (rough estimate) |
| Density | 1.0633 (rough estimate) |
| refractive index | 1.6800 (estimate) |
| storage temp. | 2-8°C |
| pka | 8.1 in 80% methylcellosolve |
| EPA Substance Registry System | Ibogaine (83-74-9) |
Description and Uses
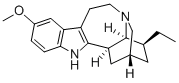
This indole alkaloid occurs in the root of Tabernanthe iboga Baill. It was first
examined in detail by Raymond-Hamet who assigned to it the empirical formula
C19H24(26)ON2, now altered to that given above. The base forms colourless
crystals from EtOH and is laevorotatory with [Q1D - 53° (EtOH). It is insoluble
in Et20, slightly so in Me2CO or CHC1 3 but dissolves freely in MeOH, EtOH or
H20. The alkaloid yields a hydrochloride, m.p. 299°C (dec.); lQlhs - 67°
(MeOH) or - 37.3° (H20). It contains one methoxyl group and gives the typical
indole reactions. On distillation with Zn dust or soda-lime it furnishes products
which are indole derivatives with the ~-position free.
In a manner similar to that of cocaine, the alkaloid potentiates the pressor
action of adrenaline and abolishes the sino-carotid reflexes. Unlike cocaine,
however, it also augments the action of tyramine and that of dl-ephedrine to a
slight extent. Vincent and Sero have reported that it inhibits the action of serum
cholinesterase.
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P264-P270-P301+P312-P330-P501 |
| Hazard Codes | F,T |
| Risk Statements | 11-23/24/25-39/23/24/25 |
| Safety Statements | 7-16-36/37-45 |
| RIDADR | 1544 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| Hazardous Substances Data | 83-74-9(Hazardous Substances Data) |