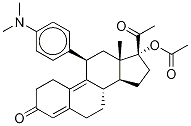
Ulipristal acetate , 98%(HPLC) , 126784-99-4
Synonym(s):
(11β)-17-(Acetyloxy)-11-[4-(dimethylamino)phenyl]-19-norpregna-4,9-diene-3,20-dione;CDB-2914
CAS NO.:126784-99-4
Empirical Formula: C30H37NO4
Molecular Weight: 475.62
MDL number: MFCD00899035
EINECS: 682-170-1
| Pack Size | Price | Stock | Quantity |
| 10MG | RMB111.20 | In Stock |
|
| 250mg | RMB349.60 | In Stock |
|
| 50MG | RMB391.20 | In Stock |
|
| 1g | RMB1219.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 183-185 ºC |
| Boiling point: | 640.1±55.0 °C(Predicted) |
| Density | 1.19 |
| storage temp. | -20°C |
| solubility | Chloroform, Ethyl Acetate, Methanol |
| pka | 5.49±0.24(Predicted) |
| form | powder |
| color | white to beige |
| optical activity | [α]/D +165 to +185°, c = 1 in dichloromethane |
Description and Uses
Ulipristal acetate is a synthetic steroid derived from 19-nor-progesterone that exerts potent progesterone receptor (PR) antagonist activity at
the transcriptional level. It is an orally administered drug indicated for
emergency contraception (i.e., postcoital contraception) up to 120 h following unprotected intercourse. Ulipristal acetate is the second oral progestogen marketed for this indication behind levonorgestrel. As PR
antagonists, both drugs are believed to act via delay of ovulation and
inhibition of follicular development. Levonorgestrel 1.5 mg is approved
for contraception up to 72 h following unprotected intercourse and is
widely accessible via prescription, and directly from clinics and community pharmacies. Prior to ulipristal acetate, the only approved postcoital
contraceptive option between 72 and 120 h was the insertion of an intrauterine device (IUD). The recommended dose of ulipristal acetate is one
30-mg tablet taken as soon as possible and no more than 120 h following
intercourse. The vast majority of PR antagonists belonging to the mifepristone (RU486) family also bind to the glucocorticoid receptor (GR)
with high affinity and thereby exert antiglucocorticoid activity.
The most common adverse effects with ulipristal acetate were headache, dysmenorrhea, nausea, abdominal pain, dizziness, fatigue, and upper abdominal pain.
Labelled Ulipristal