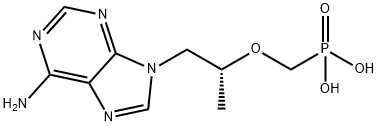
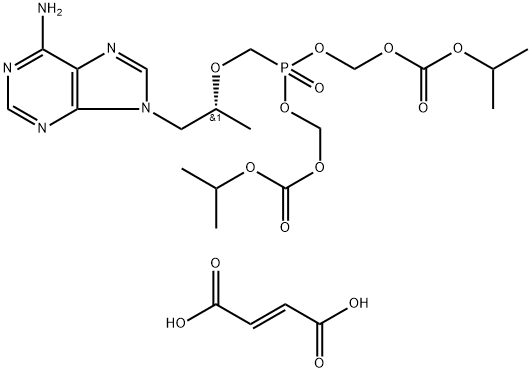
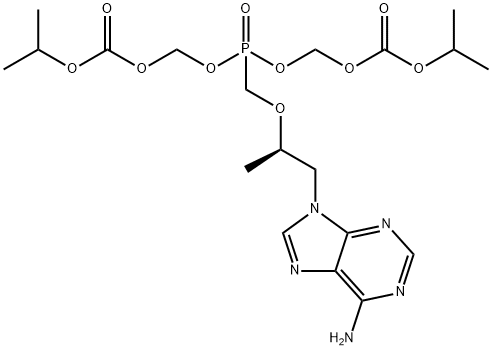
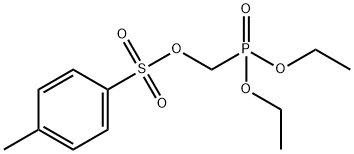
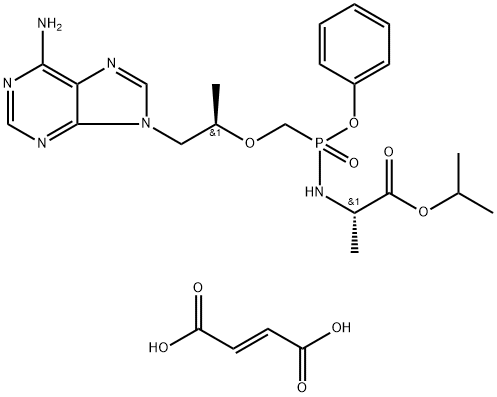
Tenofovir , ≥99% , 147127-20-6
Synonym(s):
(R)-9-(2-Phosphonomethoxypropyl)adenine;(R)-PMPA;[[(1R)-2-(6-Amino-9H-purin-9-yl)-1-methylethoxy]methyl]phosphonic acid monohydrate;1-(6-Aminopurin-9-yl) propan-2-yl-oxymethylphosphonic acid;TEN
CAS NO.:147127-20-6
Empirical Formula: C9H14N5O4P
Molecular Weight: 287.21
MDL number: MFCD07357269
EINECS: 604-571-2
| Pack Size | Price | Stock | Quantity |
| 1g | RMB27.20 | In Stock |
|
| 5G | RMB67.20 | In Stock |
|
| 25G | RMB103.20 | In Stock |
|
| 100G | RMB390.40 | In Stock |
|
| 500g | RMB1623.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 276-280°C |
| alpha | D +21° (c = 1 in 0.1M HCl) |
| Boiling point: | 616.1±65.0 °C(Predicted) |
| Density | 1.79±0.1 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Aqueous Acid (Sparingly), DMSO (Slightly, Heated), Water (Slightly, Heated) |
| pka | 2.36±0.10(Predicted) |
| form | powder |
| color | white to beige |
| optical activity | [α]/D -20 to -26°, c = 0.5 in 1 M HCl |
| Water Solubility | 13.4 mg/mL (25 ºC) |
| Merck | 14,9146 |
| InChI | InChI=1S/C9H14N5O4P/c1-6(18-5-19(15,16)17)2-14-4-13-7-8(10)11-3-12-9(7)14/h3-4,6H,2,5H2,1H3,(H2,10,11,12)(H2,15,16,17)/t6-/m1/s1 |
| InChIKey | SGOIRFVFHAKUTI-ZCFIWIBFSA-N |
| SMILES | P(CO[C@H](C)CN1C2C(N=C1)=C(N)N=CN=2)(=O)(O)O |
| CAS DataBase Reference | 147127-20-6(CAS DataBase Reference) |
Description and Uses
Tenofovir is an analog of adenosine monophosphate that has antiviral activity. It is converted by cellular enzymes to tenofovir diphosphate, an obligate chain terminator that inhibits the activity of HIV reverse transcriptase and hepatitis B virus polymerase. Tenofovir diphosphate is a weak inhibitor of mammalian DNA polymerases α and β and mitochondrial DNA polymerase γ. For in vivo and cell culture use, tenofovir is supplied as a water soluble prodrug in the form of tenofovir disoproxil (fumarate) , which increases the intracellular diphosphorylated compound >1,000-
Tenofovir is a drug used for the treatment of chronic heptatitis B as well as prevention and treatment of HIV/AIDS. It is a kind of nucleotide analog, acting as the reverse-transcriptase inhibitor (NtRTI). It inhibits the activity of HIV reverse transcriptase through competing with the natural substrate deoxyadenosine 5’-triphosphate, causing the termination of DNA chain.
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H302-H315-H319-H332-H335 |
| Precautionary statements | P261-P280-P305+P351+P338 |
| RTECS | SZ6563600 |
| HS Code | 29339900 |