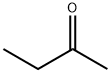
ReagentPlus ,≥99% , 78-93-3
Synonym(s):
2-Butanone;MEK;Methyl ethyl ketone;Ethyl methyl ketone;Methyl Ethyl Ketone, Butanone
CAS NO.:78-93-3
Empirical Formula: C4H8O
Molecular Weight: 72.11
MDL number: MFCD00011648
EINECS: 201-159-0
PRODUCT Properties
| Melting point: | -87 °C (lit.) |
| Boiling point: | 80 °C (lit.) |
| Density | 0.805 g/mL at 25 °C (lit.) |
| vapor density | 2.49 (vs air) |
| vapor pressure | 71 mm Hg ( 20 °C) |
| refractive index | n |
| FEMA | 2170 | 2-BUTANONE |
| Flash point: | 26 °F |
| storage temp. | 2-8°C |
| solubility | Miscible with acetone, ethanol, benzene, ether (U.S. EPA, 1985), and many other solvents,
particularly ketones and aldehydes |
| form | Solution |
| pka | 14.7 (quoted, Riddick et al., 1986) |
| color | Colorless |
| Odor | Sweet/sharp odor detectable at 2 to 85 ppm (mean = 16 ppm) |
| PH | pH(1+4, 25℃):6.0~7.0 |
| Relative polarity | 0.327 |
| explosive limit | 1.8-11.5%(V) |
| Odor Threshold | 0.44ppm |
| Odor Type | ethereal |
| Water Solubility | 290 g/L (20 ºC) |
| Merck | 14,6072 |
| JECFA Number | 278 |
| BRN | 741880 |
| Henry's Law Constant | 23.0 at 50.00 °C, 34.1 at 60.00 °C, 50.6 at 70.00 °C, 70.4 at 80.00 °C (headspace-GC, Hovorka et
al., 2002) |
| Exposure limits | TLV-TWA, PEL 590 mg/m3 (200 ppm)
(ACGIH, OSHA); STEL 885 mg/m3
(300 ppm) (ACGIH); IDLH 3000 ppm
(NIOSH). |
| Dielectric constant | 18.5(20℃) |
| Stability: | Stable. Highly flammable. Incompatible with oxidizing agents, bases, strong reducing agents. Protect from moisture. |
| LogP | 0.3 at 40℃ |
| Surface tension | 23.69mN/m at 298.15K |
| CAS DataBase Reference | 78-93-3(CAS DataBase Reference) |
| NIST Chemistry Reference | 2-Butanone(78-93-3) |
| EPA Substance Registry System | Methyl ethyl ketone (78-93-3) |
Description and Uses
2-Butanone is a stable, highly flammable chemical. It is incompatible with oxidising agents, bases, and strong reducing agents. It is a colourless liquid with a sharp, sweet odour. 2-Butanone is produced in large quantities. It is used as a solvent and nearly half of its use is in paints and other coatings because it will quickly evaporate into the air and it dissolves many substances. It is also used in glues and as a cleaning agent.
MEK is used as a solvent for various coating systems, for
example, vinyl, adhesives, nitrocellulose, and acrylic coatings.
It is used in paint removers, lacquers, varnishes, spray paints,
sealers, glues, magnetic tapes, printing inks, resins, rosins,
cleaning solutions, and for polymerization. It is found in other
consumer products, for example, household and hobby
cements, and wood-filling products. MEK is used in dewaxing
lubricating oils, the degreasing of metals, in the production of
synthetic leathers, transparent paper and aluminum foil, and as
a chemical intermediate and catalyst. It is an extraction solvent
in the processing of foodstuffs and food ingredients. MEK can
also be used to sterilize surgical and dental equipment.
In addition to its manufacture, environmental sources of
MEK include exhaust from jet and internal combustion
engines, and industrial activities such as gasification of coal. It
is found in substantial amounts in tobacco smoke. MEK is
produced biologically and has been identified as a product of
microbial metabolism. It has also been found in plants, insect
pheromones, and animal tissues, and MEK is probably a minor
product of normal mammalian metabolism. It is stable under
ordinary conditions but can form peroxides on prolonged
storage; these may be explosive.
Safety
| Symbol(GHS) |   GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225-H319-H336 |
| Precautionary statements | P210-P305+P351+P338 |
| Hazard Codes | F,Xi,T |
| Risk Statements | 11-36-66-67-39/23/24/25-23/24/25 |
| Safety Statements | 9-16-45-36/37 |
| OEB | A |
| OEL | TWA: 200 ppm (590 mg/m3), STEL: 300 ppm (885 mg/m3) |
| RIDADR | UN 1193 3/PG 2 |
| WGK Germany | 1 |
| RTECS | EL6475000 |
| Autoignition Temperature | 516 °C |
| TSCA | Yes |
| HazardClass | 3 |
| PackingGroup | II |
| HS Code | 29141200 |
| Hazardous Substances Data | 78-93-3(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats: 6.86 ml/kg (Smyth) |
| IDLA | 3,000 ppm |