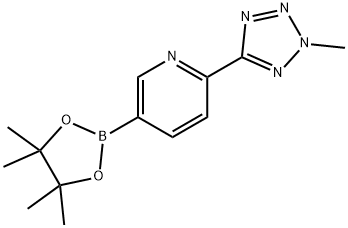
Torezolidphosphate , 99.5%, single miscellaneous <0.1% , 856867-55-5
CAS NO.:856867-55-5
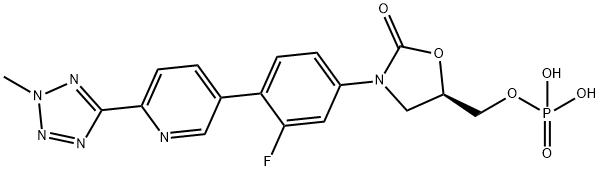
Empirical Formula: C17H16FN6O6P
Molecular Weight: 450.32
MDL number: MFCD28098176
EINECS: 1592732-453-0
| Pack Size | Price | Stock | Quantity |
| 10mg | RMB934.40 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | >235°C (dec.) |
| Boiling point: | 725.6±70.0 °C(Predicted) |
| Density | 1.75±0.1 g/cm3(Predicted) |
| storage temp. | under inert gas (nitrogen or Argon) at 2-8°C |
| solubility | DMSO (Slightly, Sonicated) |
| pka | 1.81±0.10(Predicted) |
| form | Solid |
| color | White to Off-White |
| Stability: | Hygroscopic |
| InChIKey | QCGUSIANLFXSGE-GFCCVEGCSA-N |
| SMILES | O1[C@@H](COP(O)(O)=O)CN(C2=CC=C(C3=CC=C(C4=NN(C)N=N4)N=C3)C(F)=C2)C1=O |
Description and Uses
Tedizolid phosphate was approved by the US FDA in June 2014 for treatment of acute bacterial skin and skin structure infections caused by susceptible gram-positive pathogens, including MRSA. Tedizolid phosphate was discovered by Dong-A Pharmaceuticals in South Korea and developed in the USA by Cubist Pharmaceuticals (acquired from Trius Therapeutics in 2013, became a wholly owned subsidiary of Merck in 2015). The worldwide commercialization rights for tedizolid phosphate are divided between Cubist in the USA, Canada, and EU, and Bayer in Asia–Pacific, Latin America, and Africa. This second-generation oxazolidinone prodrug is rapidly converted to the active form tedizolid in the presence of endogenous phosphatases. It inhibits bacterial protein synthesis by binding to the 23S ribosomal RNA of the 50S subunit of the ribosome, preventing formation of the 70S ribosomal initiation complex, and is 4-fold to 16-fold more potent against staphylococci and enterococci compared to linezolid. 251 With high oral bioavailability (approximately 90%) and long half-life (approximately 12 hours), tedizolid phosphate is the first oxazolidinone antibiotic which can be dosed once daily either orally or intravenously.
Tedizolid, known as TR-700, is an oral and i.v administered intracellular antibacterial drug.
Safety
| Symbol(GHS) |   GHS08,GHS09 |
| Signal word | Warning |
| Hazard statements | H400-H410-H373-H361 |
| Precautionary statements | P273-P391-P501-P260-P314-P501-P201-P202-P281-P308+P313-P405-P501-P273-P391-P501 |