HPLC≥98% , 15769-56-9
CAS NO.:15769-56-9
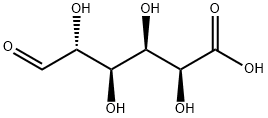
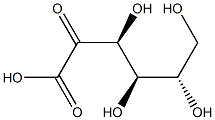
Empirical Formula: C6H10O7
Molecular Weight: 194.14
MDL number: MFCD30533975
EINECS: 239-860-9
| Pack Size | Price | Stock | Quantity |
| 10mg | RMB1360.00 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| form | Solid |
| color | White to off-white |
Description and Uses
Glucuronic acid is a sugar acid derived from glucose, with its sixth carbon atom oxidized to a carboxylic acid. In living beings, this primary oxidation occurs with UDP-α-D-glucose (UDPG), not with the free sugar.
Glucuronic acid, like its precursor glucose, can exist as a linear (carboxo-)aldohexose (<1%), or as a cyclic hemiacetal (furanose or pyranose). Aldohexoses such as D-glucose are capable of forming two furanose forms (α and β) and two pyranose forms (α and β). By the Fischer convention, glucuronic acid has two stereoisomers (enantiomers), D- and L-glucuronic acid, depending on its configuration at C-5. Most physiological sugars are of the D-configuration. Due to ring closure, cyclic sugars have another asymmetric carbon atom (C-1), resulting in two more stereoisomers, named anomers. Depending on the configuration at C-1, there are two anomers of glucuronic acid, α- and β-form. In β-D-glucuronic acid the C-1 hydroxy group is on the same side of the pyranose ring as the carboxyl group. In the free sugar acid, the β-form is prevalent (~64%), whereas in the organism, the α-form UDP-α-D-glucuronic acid (UDPGA) predominates.
Guluronic acid (G2013), one of the organic building blocks of hyaluronic acid, is a nonsteroidal anti-inflammatory agent has favorable anti-inflammatory effect[1][2].