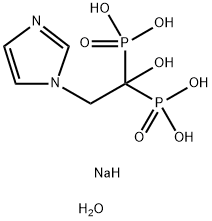
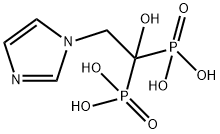
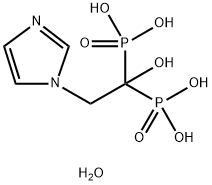
Zoledronate disodium tetrahydrate , 98%+ , 165800-07-7
CAS NO.:165800-07-7
Empirical Formula: C5H13N2NaO8P2
Molecular Weight: 314.1
MDL number: MFCD07357645
PRODUCT Properties
| Melting point: | 305-307°C |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | Methanol (Slightly, Heated, Sonicated), Water (Slightly, Heated) |
| form | Solid |
| color | White to Off-White |
Description and Uses
Zoledronate was introduced in Canada as a lyophilized powder in vials for infusion as solution in the treatment of tumor-induced hypercalcemia. This third-generation bisphosphonate can be obtained by reaction of 2-(1-imidazolyl)acetic acid hydrochloride with phosphorous trichloride followed by hydrolysis in concentrated hydrochloric acid, Zoledronate originated from a medicinal chemistry program started at Novartis, Switzerland in 1986 and aimed at discovering a follow-up to pamidronate for the treatment of osteoporosis and metastatic bone disease. It has highly potent inhibitory effect on hypercalcemia induced by vitamin Ds both in vitro in mouse calvarial cultures and in viva in the thyroparathyroidectomized rat. It is respectively 100 and 850-fold more potent than pamidronate in the former and latter tests. Clinically, it was demonstrated that zoledronate achieved normalized serum calcium concentrations in a significantly higher percentage of cancer patients compared to pamidronate. The 5 minute infusion with zoledronate was more convenient than the 2 hour infusion necessary with pamidronate.
A biphosphonate bone resorption inhibitor.