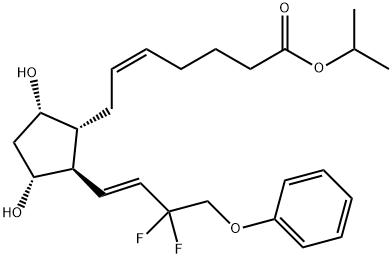
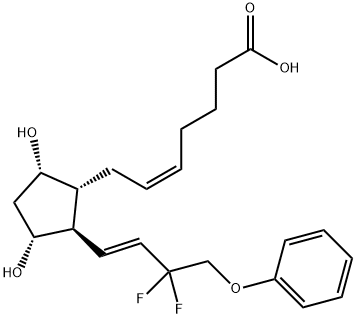
Tafluprost , 97% , 209860-87-7
CAS NO.:209860-87-7
Empirical Formula: C25H34F2O5
Molecular Weight: 452.53
MDL number: MFCD08062150
EINECS: 234-199-2
| Pack Size | Price | Stock | Quantity |
| 100mg | RMB1219.20 | In Stock |
|
| 250mg | RMB1828.80 | In Stock |
|
| 1g | RMB4571.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Boiling point: | 552.9±50.0 °C(Predicted) |
| Density | 1.186 |
| storage temp. | 2-8°C |
| solubility | soluble in Chloroform, DMSO, Ethyl Acetate |
| form | Clear, colorless to slightly yellow oil |
| pka | 14.48±0.70(Predicted) |
| color | Colorless to light yellow |
| optical activity | [α]/D +20 to +25°, c =1 in chloroform-d |
| InChIKey | WSNODXPBBALQOF-VEJSHDCNSA-N |
| SMILES | C(OC(C)C)(=O)CCC/C=C\C[C@H]1[C@@H](O)C[C@@H](O)[C@@H]1/C=C/C(F)(F)COC1=CC=CC=C1 |
Description and Uses
Glaucoma is second only to cataracts as a causative factor of blindness.
By 2010, it is estimated that approximately 60 million people worldwide
will be afflicted by glaucoma, so effective treatments should garner a large market.
PG analogs have been widely used for lowering IOP by increasing uveoscleral outflow through agonism of the prostanoid FP
receptor, and currently marketed versions include latanoprost, unoprostone isopropyl ester, bimatoprost, and travoprost.
Compared to the carboxylic acid of latanaprost
(Ki=4.7 nM), the carboxylic acid of tafluprost displayed a 10-fold greater
affinity for the prostanoid FP receptor (Ki=0.4 nM). The synthesis of
tafluprost begins with a Wittig condensation of the protected bicyclic
lactone carbaldehyde with a dimethyl phosphonate ketone derivative.
Compared to the carboxylic acid of latanaprost (Ki 4.7 nM), the carboxylic acid of tafluprost displayed a 10-fold greater affinity for the prostanoid FP receptor (Ki 0.4 nM). The synthesis of tafluprost begins with a Wittig condensation of the protected bicyclic lactone carbaldehyde with a dimethyl phosphonate ketone derivative. The bottom appendage is then completed by the fluorination of the ketone with morpholino-sulfur trifluoride. Hydrolysis of the benzoate ester protecting group liberates the hydroxy group, and reduction of the lactone is accomplished with aluminum hydride to generate the lactol. Condensation of this intermediate with the phosphonium salt of the acid side chain generates the free acid, or active ingredient, which is subsequently esterified with 2-iodopropane in the presence of DBU.
.
Tafluprost is a novel prostanoid used in the treatment of glaucoma and is the first prostanoid to be released in a preservative free-formula.
Safety
| Symbol(GHS) |   GHS08,GHS07 |
| Signal word | Danger |
| Hazard statements | H372-H360-H319-H413-H370-H302 |
| Precautionary statements | P264-P280-P305+P351+P338-P337+P313P-P260-P264-P270-P307+P311-P321-P405-P501-P264-P270-P301+P312-P330-P501-P260-P264-P270-P314-P501 |