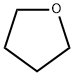
Tetrahydrofuran , Waterless grade, ≥99.9%, no stabilizer , 109-99-9
Synonym(s):
THF;Tetrahydrofuran;Oxolane;Butylene oxide;PTHF
CAS NO.:109-99-9
Empirical Formula: C4H8O
Molecular Weight: 72.11
MDL number: MFCD00005356
EINECS: 203-726-8
PRODUCT Properties
| Melting point: | -108°C |
| Boiling point: | 66 °C |
| Density | 0.887 g/mL at 20 °C |
| vapor density | 2.5 (vs air) |
| vapor pressure | <0.01 mm Hg ( 25 °C) |
| refractive index | n |
| Flash point: | >230 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | water: soluble |
| form | Liquid |
| Specific Gravity | 0.89 |
| color | <10(APHA) |
| Odor | Ethereal, detectable at 2 to 50 ppm |
| PH Range | 7 |
| Relative polarity | 0.207 |
| PH | 7-8 (200g/l, H2O, 20℃) |
| explosive limit | 1.5-12.4%(V) |
| Water Solubility | miscible |
| FreezingPoint | -108℃ |
| Sensitive | Air Sensitive & Hygroscopic |
| λmax | λ: 245 nm Amax: ≤0.26 λ: 275 nm Amax: ≤0.046 λ: 315 μm Amax: ≤0.0044 |
| Merck | 14,9211 |
| BRN | 102391 |
| Henry's Law Constant | 1.54 (static headspace-GC, Welke et al., 1998) |
| Exposure limits | TLV-TWA 200 ppm (590 mg/m3) (ACGIH,
MSHA, and OSHA); STEL 250 ppm
(ACGIH); IDLH 20,000 ppm (NIOSH). |
| Dielectric constant | 11.6(-70℃) |
| Stability: | Stable. Incompatible with halogens, strong oxidizing agents, strong reducing agents, strong bases, oxygen. May generate explosive peroxides in storage if in contact with air. Highly flammable. Store at room temperature under nitrogen. Hazardous polymerisation may occur. Light sensitive. May contain 2,6-di-tertbutyl-4-methylphenol (BHT) as a s |
| InChIKey | WYURNTSHIVDZCO-UHFFFAOYSA-N |
| LogP | 0.45 at 25℃ |
| Surface tension | 27.31mN/m at 293.15K |
| CAS DataBase Reference | 109-99-9(CAS DataBase Reference) |
| IARC | 2B (Vol. 119) 2019 |
| NIST Chemistry Reference | Furan, tetrahydro-(109-99-9) |
| EPA Substance Registry System | Tetrahydrofuran (109-99-9) |
| Absorption | ≤0.0044 at 315nm ≤0.005 at 350nm ≤0.005 at 400nm ≤0.02 at 300nm ≤0.046 at 275nm ≤0.18 at 250nm ≤0.26 at 245nm ≤1.0 at 212nm |
Description and Uses
Tetrahydrofuran (THF) is a colorless, volatile liquid with an ethereal or acetonelike smell and is miscible in water and most organic solvents.It is highly flammable and may thermally decompose to carbon monoxide and carbon dioxide. Prolonged storage in contact with air and in the absence of an antioxidant may cause THF to decompose into explosive peroxides.
Tetrahydrofuran is used in the manufacture of polymers as well as agricultural, pharmaceutical, and commodity chemicals. Manufacturing activities commonly occur in closed systems or under engineering controls that limit worker exposure and release to the environment. THF is also used as a solvent (e.g., pipe fitting) that may result in more significant exposures when used in confined spaces without sufficient ventilation. Although THF is naturally present in coffee aroma, floured chickpeas, and cooked chicken, natural exposures are not anticipated to pose a significant hazard.
Tetrahydrofuran is used as a solvent forresins, vinyls, and high polymers; as a Grignardreaction medium for organometallic,and metal hydride reactions; and in the synthesisof succinic acid and butyrolactone.
Safety
| Symbol(GHS) |    GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H225-H302-H319-H335-H336-H351 |
| Precautionary statements | P201-P202-P210-P301+P312-P305+P351+P338-P308+P313 |
| Hazard Codes | Xi,F,Xn |
| Risk Statements | 36/37/38-36/37-19-11-40 |
| Safety Statements | 26-36-33-29-16-46-37-13 |
| RIDADR | UN 2924 3/PG 2 |
| OEB | A |
| OEL | TWA: 200 ppm (590 mg/m3), STEL: 250 ppm (735 mg/m3) |
| WGK Germany | 1 |
| RTECS | MD0916000 |
| F | 3-10-23 |
| Autoignition Temperature | 610 °F |
| TSCA | Yes |
| HazardClass | 3 |
| PackingGroup | II |
| HS Code | 29321100 |
| Hazardous Substances Data | 109-99-9(Hazardous Substances Data) |
| Toxicity | LD50 oral (rat) 2880 mg/kg LC50 inhal (rat) 21,000 ppm (3 h) PEL (OSHA) 200 ppm (590 mg/m3) TLV-TWA (ACGIH) 200 ppm (590 mg/m3) STEL (ACGIH) 250 ppm (737 mg/m3) |
| IDLA | 2,000 ppm [10% LEL] |