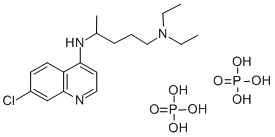
ChloroquinePhosphate , 10mMinWater , 50-63-5
Synonym(s):
N4-(7-Chloro-4-quinolinyl)-N1,N1-dimethyl-1,4-pentanediamine diphosphate salt;Chloroquine diphosphate salt;N4-(7-chloroquinolin-4-yl)-N1,N1-diethylpentane-1,4-diamine diphosphate
CAS NO.:50-63-5
Empirical Formula: C18H32ClN3O8P2
Molecular Weight: 515.86
MDL number: MFCD00069852
EINECS: 200-055-2
| Pack Size | Price | Stock | Quantity |
| 1ml | RMB159.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 200 °C (dec.) (lit.) |
| vapor pressure | <0.0000001 kPa ( 25 °C) |
| storage temp. | protect from light |
| solubility | H2O: 50 mg/mL, clear |
| form | solid |
| pka | pKa 8.10(H2O
t = 20
c = 0.0025) (Uncertain) |
| color | White |
| PH | pH(100g/l, 25℃) : 3.8~4.3 |
| biological source | synthetic |
| Water Solubility | Soluble in water |
| Merck | 14,2163 |
| BRN | 4223142 |
| BCS Class | 1 |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in distilled water may be stored at -20° for up to 3 months. |
| InChIKey | QKICWELGRMTQCR-UHFFFAOYSA-N |
| CAS DataBase Reference | 50-63-5(CAS DataBase Reference) |
| EPA Substance Registry System | Chloroquine diphosphate (50-63-5) |
Description and Uses
Chloroquine is an aminoquinoline that is an inhibitor of autophagy and has antimalarial, anti-inflammatory, anticancer, and antiviral activities. Chloroquine inhibits autophagosome-lysosome fusion in HeLa cells when used at a concentration of 100 μM. It is active against the chloroquine-sensitive GC03 strain of P. falciparum (IC50 = 29.2 nM) but has decreased activity against mutant pfcrt P. falciparum (IC50s = 100-150 nM). Chloroquine prevents infection by severe acute respiratory coronavirus 2 (SARS-CoV-2) in Vero cells (EC50 = 1.13 μM) but does not inhibit SARS-CoV replication in the lungs in a mouse model of SARS-CoV infection. It inhibits the growth of human SSC25 and CAL 27 oral squamous cell carcinoma cells (IC50s = 29.9 and 17.3 μM, respectively), as well as A498, SN12C, RXF 393, and 769-P renal cancer cells (IC50s = 16, 62, 81, and 25 μM, respectively). It reduces tumor growth in a CAL 27 mouse xenograft model and a 4T1 mouse allograft model when administered at a dose of 50 mg/kg. Formulations containing chloroquine have been used in the prevention of malaria, as well as the treatment of rheumatoid arthritis and systemic lupus erythematosus (SLE), and have been associated with cardiotoxicity and myopathy.
An antimalarial compound
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P264-P270-P301+P312-P501 |
| Hazard Codes | Xn |
| Risk Statements | 22-40-20/21/22 |
| Safety Statements | 22-24/25-36 |
| RIDADR | 1544 |
| WGK Germany | 3 |
| RTECS | VB2450000 |
| F | 10 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| HS Code | 2933492250 |
| Toxicity | LD50 oral in rat: 623mg/kg |