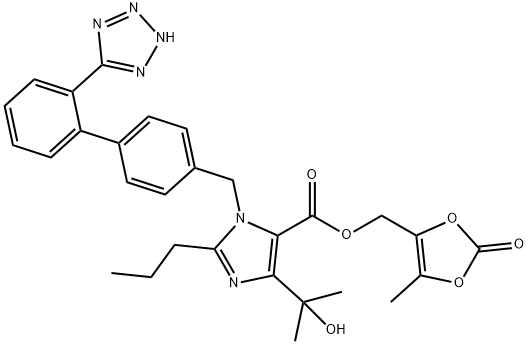
Olmesartan Medoxomil , ≥98% , 144689-63-4
Synonym(s):
4-(1-Hydroxy-1-methylethyl)-2-propyl-1-[[2′-(2H-tetrazol-5-yl)[1,1′-biphenyl]-4-yl]methyl]-1H-imidazole-5-carboxylic acid (5-methyl-2-oxo-1,3-dioxol-4-yl)methyl ester;CS-866;Olmesartan medoxomil
CAS NO.:144689-63-4
Empirical Formula: C29H30N6O6
Molecular Weight: 558.59
MDL number: MFCD00944911
EINECS: 604-433-1
| Pack Size | Price | Stock | Quantity |
| 10MG | RMB408.80 | In Stock |
|
| 1g | RMB424.00 | In Stock |
|
| 50MG | RMB520.00 | In Stock |
|
| 5g | RMB1480.00 | In Stock |
|
| 250MG | RMB6639.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 180°C |
| Boiling point: | 804.2±75.0 °C(Predicted) |
| Density | 1.38±0.1 g/cm3(Predicted) |
| Flash point: | 180°C |
| storage temp. | 2-8°C |
| solubility | DMSO: soluble20mg/mL, clear |
| pka | 4.15±0.10(Predicted) |
| form | powder |
| color | white to beige |
| Decomposition | 180 ºC |
| InChIKey | UQGKUQLKSCSZGY-UHFFFAOYSA-N |
| SMILES | C1(CCC)N(CC2=CC=C(C3=CC=CC=C3C3=NNN=N3)C=C2)C(C(OCC2=C(C)OC(=O)O2)=O)=C(C(O)(C)C)N=1 |
| CAS DataBase Reference | 144689-63-4(CAS DataBase Reference) |
Description and Uses
Olmesartan medoxomil was launched in the US as benicar(R), an orally administered treatment for hypertension. Olmesartan, is a new selective and competitive nonpeptide angiotensin II type 1 receptor antagonist and potently inhibits the Ang.ll-induced pressor responses. The drug competitively inhibited binding of [125I1]-All to AT1 receptors in bovine adrenal cortical membranes, but had no effect on binding to AT2 receptors in bovine cerebellar membranes. In comparative clinical studies in patients with essential hypertension, olmesartan reduced sitting cuff diastolic blood pressure significantly more than losartan, valdesartan and ibesartan, while reductions in systolic blood pressure were similar for all treatments. Olmesartan medoxomil was also shown to reduce blood pressure significantly more effectively than losartan and the ACE inhibitor captopril and as effectively as the pbloker atenolol.
Olmesartan medoxomil is an angiotensin II receptor antagonist used to treat high blood pressure. Olmesartan works by blocking the binding of angiotensin II to the AT1 receptors in vascular muscle. By blocking the binding rather than the synthesis of angiotensin II, olmesartan inhibits the negative regulatory feedback on renin secretion.
Olmesartan medoxomil is a pro-drug that is de-esterified to the active metabolite, olmesartan. Olmesartan has a dual method of elimination, with about 60% eliminated by the liver and the remainder by the kidney. In situations of impaired renal or hepatic function, the alternative excretion pathway can compensate for the compromised one. Olmesartan is not metabolized by the cytochrome P450 enzyme system and therefore has a low potential for metabolic drug interactions, a feature that may be of importance when treating patients on multiple drug regimens, such as the elderly. Olmesartan is well tolerated and has an excellent safety profile that is comparable to that of placebo. In addition, olmesartan provides 24-h blood pressure control with a once-daily dosing. In head-to-head studies, olmesartan delivered superior blood pressure reduction when compared with other angiotensin-II receptor antagonists at their recommended doses.
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H302+H312+H332 |
| Precautionary statements | P261-P264-P280-P301+P312-P302+P352+P312-P304+P340+P312 |
| RTECS | NI4014200 |
| HS Code | 2934990002 |