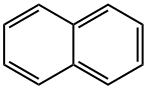
Naphthalene , StandardforGC,≥99.5%(GC) , 91-20-3
Synonym(s):
Naphthalene
CAS NO.:91-20-3
Empirical Formula: C10H8
Molecular Weight: 128.17
MDL number: MFCD00001742
EINECS: 202-049-5
PRODUCT Properties
| Melting point: | 80-82 °C (lit.) |
| Boiling point: | 218 °C (lit.) |
| Density | 0.99 |
| bulk density | 550-600mg/m3 |
| vapor density | 4.4 (vs air) |
| vapor pressure | 0.03 mm Hg ( 25 °C) |
| refractive index | 1.5821 |
| Flash point: | 174 °F |
| storage temp. | Store below +30°C. |
| solubility | methanol: soluble50mg/mL, clear, colorless |
| form | Faint beige to brown to salmon red powder |
| color | White to almost white |
| Specific Gravity | 1.145 |
| Odor | at 10.00 % in benzyl benzoate. pungent dry tarry |
| Odor Type | pungent |
| explosive limit | 0.9-5.9%(V) |
| Water Solubility | 30 mg/L (25 ºC) |
| Merck | 14,6370 |
| BRN | 1421310 |
| Henry's Law Constant | 5.64 at 25 °C (thermodynamic method-GC/UV spectrophotometry, Altschuh et al., 1999) |
| Exposure limits | TLV-TWA 10 ppm (~50 mg/m3) (ACGIH,
MSHA, and OSHA); STEL 15 ppm
(~75 mg/m3) (ACGIH); IDLH 500 ppm. |
| Dielectric constant | 2.5(85℃) |
| InChI | 1S/C10H8/c1-2-6-10-8-4-3-7-9(10)5-1/h1-8H |
| InChIKey | UFWIBTONFRDIAS-UHFFFAOYSA-N |
| SMILES | c1ccc2ccccc2c1 |
| LogP | 3.7 at 25℃ |
| CAS DataBase Reference | 91-20-3(CAS DataBase Reference) |
| IARC | 2B (Vol. 82) 2002 |
| NIST Chemistry Reference | Naphthalene(91-20-3) |
| EPA Substance Registry System | Naphthalene (91-20-3) |
Description and Uses
Naphthalene occurs as transparent prismatic plates also available as white scales, powder balls, or cakes with a characteristic mothball or strong coal tar and aromatic odour. It is sparingly soluble in water but soluble in methanol/ethanol and very soluble in ether. Naphthalene is a commercially important aromatic hydrocarbon. Naphthalene occurs as a white solid or powder. Naphthalene occurs in coal tar in large quantities and is easily isolated from this source in pure condition. It volatilises and sublimes at room temperature above the melting point. The primary use for naphthalene is in the production of phthalic anhydride, also of carbamate insecticides, surface active agents and resins, as a dye intermediate, as a synthetic tanning agent, as a moth repellent, and in miscellaneous organic chemicals. Naphthalene is used in the production of phthalic anhydride; it is also used in mothballs. Naphthalene is also used in the manufacture of phthalic and anthranilic acids to make indigo, indanthrene, and triphenyl methane dyes, for synthetic resins, lubricant, celluloid, lampblack, smokeless powder, and hydronaphthalenes. Naphthalene is also used in dusting powders, lavatory deodorant discs, wood preservatives, fungicide, and as an insecticide. It has been used as an intestinal antiseptic and vermicide and in the treatment of pediculosis and scabies.
In addition to oxidation and reduction reactions, naphthalene readily undergoes substitutionreactions such as nitration, halogenation, sulfonation, and acylation to produce a varietyof other substances, which are used in the manufacture of dyes, insecticides, organic solvents,and synthetic resins. The principal use of naphthalene is for the production of phthalic anhydride,C8H4O3.
Naphthalene is catalytically oxidized to phthalic anhydride: 2C10H8 + 9O2 → 2C4H8O3 +4CO2 + 4H2O using metal oxide catalysts. Phthalic anhydride is used to produce plastics,phthalate plasticizers, insecticides, pharmaceuticals, and resins. Sulfonation of naphthalene withsulfuric acid produces naphthalenesulfonic acids, which are used to produce naphthalene sulfonates.Naphthalene sulfonates are used in various formulations as concrete additives, gypsumboard additives, dye intermediates, tanning agents, and polymeric dispersants. Naphthalene isused to produce carbamate insecticides such as carbaryl, which is a wide-spectrum, generalpurposeinsecticide.
Safety
| Symbol(GHS) |     GHS02,GHS07,GHS08,GHS09 |
| Signal word | Warning |
| Hazard statements | H228-H302-H351-H410 |
| Precautionary statements | P202-P210-P240-P273-P301+P312-P308+P313 |
| PPE | Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges |
| Hazard Codes | Xn,N,F,T |
| Risk Statements | 22-40-50/53-67-65-38-11-39/23/24/25-23/24/25-52/53-20 |
| Safety Statements | 36/37-46-60-61-62-45-16-7-33-25-9 |
| RIDADR | UN 1334 4.1/PG 3 |
| OEB | A |
| OEL | TWA: 10 ppm (50 mg/m3), STEL: 15 ppm (75 mg/m3) |
| WGK Germany | 3 |
| RTECS | QJ0525000 |
| Autoignition Temperature | 978 °F |
| TSCA | TSCA listed |
| HazardClass | 4.1 |
| PackingGroup | III |
| HS Code | 29029010 |
| Storage Class | 4.1B - Flammable solid hazardous materials |
| Hazard Classifications | Acute Tox. 4 Oral Aquatic Acute 1 Aquatic Chronic 1 Carc. 2 Flam. Sol. 2 |
| Hazardous Substances Data | 91-20-3(Hazardous Substances Data) |
| Toxicity | Acute oral LD50 for guinea pigs 1,200 mg/kg, mice 533 mg/kg, rats 1,250 mg/kg (quoted, RTECS, 1985). |
| IDLA | 250 ppm |