Lenalidomide , ≥99% , 191732-72-6
Synonym(s):
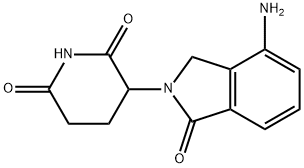
Lenalidomide;3-(4-Amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione;3-(4-Amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl)-2,6-piperidinedione;3-(4-Amino-1-oxoisoindolin-2-yl)piperidin-2,6-dione;1-Oxo-4-amino-2-(2,6-dioxopiperidin-3-yl)isoindole
CAS NO.:191732-72-6
Empirical Formula: C13H13N3O3
Molecular Weight: 259.26
MDL number: MFCD18064659
EINECS: 691-297-1
| Pack Size | Price | Stock | Quantity |
| 50MG | RMB23.20 | In Stock |
|
| 250MG | RMB28.00 | In Stock |
|
| 1G | RMB56.80 | In Stock |
|
| 5g | RMB200.80 | In Stock |
|
| 25g | RMB799.20 | In Stock |
|
| 100g | RMB2639.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 265-268 °C |
| Boiling point: | 614.0±55.0 °C(Predicted) |
| Density | 1.460±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Soluble in DMSO (up to 30 mg/ml) |
| pka | 10.75±0.40(Predicted) |
| form | solid |
| color | White |
| Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
| InChI | InChI=1S/C13H13N3O3/c14-9-3-1-2-7-8(9)6-16(13(7)19)10-4-5-11(17)15-12(10)18/h1-3,10H,4-6,14H2,(H,15,17,18) |
| InChIKey | GOTYRUGSSMKFNF-UHFFFAOYSA-N |
| SMILES | N1C(=O)CCC(N2CC3=C(C2=O)C=CC=C3N)C1=O |
| CAS DataBase Reference | 191732-72-6(CAS DataBase Reference) |
Description and Uses
Lenalidomide is a kind of antitumor drugs that developed by American biological pharmaceutical companies. Its chemical structure is similar with thalidomide. It differing in the presence of an amino moiety in the 4-position and removal of one of the carbonyls of the phthaloyl ring. This derivative evolved from a structural-based effort to eliminate the adverse effects (somnolence, neuropathy, and teratogenicity) of thalidomide while maintaining or enhancing the appealing attributes. It has many functions such as anti-tumor, immune regulation and anti-angiogenesis. It can inhibit the secretion of inflammatory cytokines, and increase the secretion of peripheral blood mononuclear anti-inflammatory cytokines. Vitro tests show that lenalidomide can inhibit the hyperplasia of some cell lines such as namalwa cell. It can inhibit the growth of patients’ multiple myeloma cells and MM1S cell. In addition, lenalidomide also can inhibit the expression of oxidase-2 (COX-2), but it has no effect on COX-1. Two multicenter randomized double-blind placebo-controlled clinical studies evaluate the safety and curative effect of lenalidomide that is used for multiple myeloma. The primary efficacy end point of the studies is time to progression (TTP). The interim analysis shows that TTP of the combination group is significantly superior to dexamethasone group. Recent clinical research results show that lenalidomide not only has curative effect on treating MDS and MM, but also on treating myeloma, leukemia, metastatic renal cell carcinoma, solid tumor, idiopathic generalized amyloidosis and systemic bone marrow fibrosis disease with marrow unripe.
Lenalidomide is a thalidomide analog known to have immunomodulatory properties. Lenalidomide inhibits TNF-alpha production, stimulates T cells, reduces serum levels of the cytokines vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF), and inhibits angiogenesis. This agent also promotes G1 cell cycle arrest and apoptosis of malignant cells. It is used in the therapy of multiple myeloma.
Safety
| Symbol(GHS) |  GHS08 |
| Signal word | Warning |
| Hazard statements | H361d-H373 |
| Precautionary statements | P202-P260-P280-P308+P313-P405-P501 |
| Risk Statements | 20/21/22 |
| Safety Statements | 36/37 |
| HazardClass | IRRITANT |
| HS Code | 29339900 |
| Hazardous Substances Data | 191732-72-6(Hazardous Substances Data) |