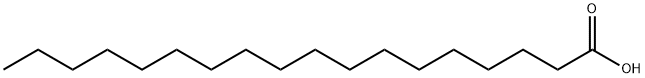
Stearic acid , C18:50% , 57-11-4
Synonym(s):
Stearic acid;Octadecanoic acid;STA;C18:0;Cetylacetic acid
CAS NO.:57-11-4
Empirical Formula: C18H36O2
Molecular Weight: 284.48
MDL number: MFCD00002752
EINECS: 266-928-5
| Pack Size | Price | Stock | Quantity |
| 1kg | RMB47.20 | In Stock |
|
| 5kg | RMB183.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 67-72 °C (lit.) |
| Boiling point: | 361 °C (lit.) |
| bulk density | 400-500kg/m3 |
| Density | 0.845 g/cm3 |
| vapor pressure | 1 mm Hg ( 173.7 °C) |
| FEMA | 3035 | STEARIC ACID |
| refractive index | 1.4299 |
| Flash point: | >230 °F |
| storage temp. | Store below +30°C. |
| solubility | Practically insoluble in water, soluble in ethanol (96 per cent) and in light petroleum (bp: 50-70 °C). |
| form | powder |
| pka | pKa 5.75±0.00(H2O
t = 35) (Uncertain) |
| Specific Gravity | 0.84 (80℃) |
| color | White |
| Odor | odorless mild fatty |
| Odor Type | odorless |
| biological source | palm oil |
| Water Solubility | 0.1-1 g/100 mL at 23 ºC |
| JECFA Number | 116 |
| Merck | 14,8804 |
| BRN | 608585 |
| Dielectric constant | 2.3(22℃) |
| Exposure limits | ACGIH: TWA 10 mg/m3; TWA 3 mg/m3 |
| InChIKey | QIQXTHQIDYTFRH-UHFFFAOYSA-N |
| LogP | 8.22 |
| CAS DataBase Reference | 57-11-4(CAS DataBase Reference) |
| NIST Chemistry Reference | Octadecanoic acid(57-11-4) |
| EPA Substance Registry System | Stearic acid (57-11-4) |
Description and Uses
Stearic acid is a long-chain saturated fatty acid. It is a major component of cocoa butter and has also been found in beef fat and vegetable oils. Unlike many long-chain saturated fatty acids, dietary stearic acid does not induce hypercholesterolemia or raise LDL-cholesterol.
Generally applications of stearic acid exploit its bifunctional character, with a polar head group that can be attached to metal cations and a nonpolar chain that confers solubility in organic solvents. The combination leads to uses as a surfactant and softening agent. Stearic acid undergoes the typical reactions of saturated carboxylic acids, notably reduction to stearyl alcohol, and esterification with a range of alcohols.
Soaps , cosmetics , detergents
Stearic acid is mainly used in the production of detergents, soaps, and cosmetics such as shampoos and shaving cream products. Soaps are not made directly from stearic acid, but indirectly by saponification of triglycerides consisting of stearic acid esters. Esters of stearic acid with ethylene glycol; glycol stearate and glycol distearate, are used to produce a pearly effect in shampoos, soaps, and other cosmetic products. They are added to the product in molten form and allowed to crystallize under controlled conditions. Detergents are obtained from amides and quaternary alkylammonium derivatives of stearic acid.
Lubricants , softening and release agents
In view of the soft texture of the sodium salt, which is the main component of soap, other salts are also useful for their lubricating properties. Lithium stearate is an important component of grease. The stearate salts of zinc, calcium, cadmium, and lead are used to soften PVC. Stearic acid is used along with castor oil for preparing softeners in textile sizing. They are heated and mixed with caustic potash or caustic soda. Related salts are also commonly used as release agents, e.g. in the production of automobile tires.
Niche uses
Being inexpensively available and chemically benign, stearic acid finds many niche applications. When making plaster castings from a plaster piece mold or waste mold and when making the mold from a shellacked clay original. In this use, powdered stearic acid is mixed in water and the suspension is brushed onto the surface to be parted after casting. This reacts with the calcium in the plaster to form a thin layer of calcium stearate which functions as a release agent. When reacted with zinc it forms zinc stearate which is used a lubricant for playing cards (fanning powder) to ensure a smooth motion when fanning. In compressed confections, it is used as a lubricant to keep the tablet from sticking to the die.
Fatty acids are classic components of candle - making. Stearic acid is used along with simple sugar or corn syrup as a hardener in candies.
Stearic acid is used to produce dietary supplements.
In fire works, stearic acid is often used to coat metal powders such as aluminium and iron. This prevents oxidation, allowing compositions to be stored for a longer period of time. Stearic acid is a common lubricant during injection molding and pressing of ceramic powders. It is also used as a mold release for foam latex that is baked in stone molds. .
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P280a-P304+P340-P305+P351+P338-P405-P501a |
| Hazard Codes | Xi,F |
| Risk Statements | 38-36/37/38-11 |
| Safety Statements | 37/39-26-16 |
| WGK Germany | 3 |
| RTECS | WI2800000 |
| Autoignition Temperature | 395 °C |
| TSCA | Yes |
| HS Code | 38231100 |
| Hazardous Substances Data | 57-11-4(Hazardous Substances Data) |
| Toxicity | LD50 i.v. in mice, rats: 23±0.7, 21.5±1.8 mg/kg, L. Or, A. Wretlind, Acta Pharmacol. Toxicol. 18, 141 (1961) |