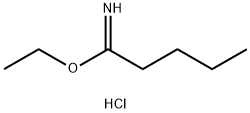
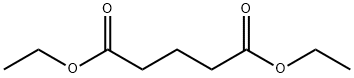
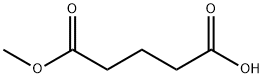
Glutarimide , >98.0%(HPLC) , 1121-89-7
Synonym(s):
2,6-Piperidinedione;NSC 58190
CAS NO.:1121-89-7
Empirical Formula: C5H7NO2
Molecular Weight: 113.11
MDL number: MFCD00006670
EINECS: 214-340-4
| Pack Size | Price | Stock | Quantity |
| 1g | RMB24.00 | In Stock |
|
| 5G | RMB76.00 | In Stock |
|
| 25G | RMB328.00 | In Stock |
|
| 100g | RMB1233.60 | In Stock |
|
| 500g | RMB4335.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 155-157 °C (lit.) |
| Boiling point: | 211.82°C (rough estimate) |
| Density | 1.2416 (rough estimate) |
| refractive index | 1.4200 (estimate) |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | soluble in Chloroform, DCM |
| form | Crystals or Crystalline Flakes |
| pka | pKa 11.4 (Uncertain) |
| color | White |
| Water Solubility | Soluble in water, hot ethanol and boiling benzene. Insoluble in ether. |
| BRN | 110052 |
| InChI | InChI=1S/C5H7NO2/c7-4-2-1-3-5(8)6-4/h1-3H2,(H,6,7,8) |
| InChIKey | KNCYXPMJDCCGSJ-UHFFFAOYSA-N |
| SMILES | N1C(=O)CCCC1=O |
| CAS DataBase Reference | 1121-89-7(CAS DataBase Reference) |
| EPA Substance Registry System | Glutarimide (1121-89-7) |
Description and Uses
Glutarimide, also known as Piperidine-2,6-dione or 2,6-Piperidinedione, is a versatile and commonly used organic reagent, classified in pharmacology as a protein synthesis inhibitor compound. It is also an important component of the anti-cancer drug thalidomide. The study reported the synthesis of triazoloquinoxaline derivative (Compound 15), a new potent anticancer drug containing a glutarimide molecule, based on the CRBN binding property of the glutarimide molecule in thalidomide molecule and the cytotoxic activity of triazole-quinoxaline. The IC50s were 9.81 ± 0.7, 15.49 ± 1.2 and 10.09 ± 0.9 μM for hepatocellular carcinoma (HepG2), prostate cancer (PC3) and breast cancer (MCF-7), respectively. It was superior to thalidomide in decreasing NF-κB P65 levels in HepG-2 cells[1].
Glutarimide acts as an inhibitor of protein synthesis. Further, it is used as a reactant for thionations and biocatalytic asymmetric synthesis of sitagliptin production. It is also employed in the generation of beta-adrenoceptor ligands, enantioselective synthesis of securinega alkaloids and alfa-fluoro-alfa amino amides. In addition to this, it is used in intramolecular amidocyclopropanation reactions.

![N-[5-(Phenylamino)-2,4-pentadienylidene]aniline monohydrochloride](https://img.chemicalbook.com/CAS/GIF/1497-49-0.gif)