Ezetimibe , ≥99% , 163222-33-1
Synonym(s):
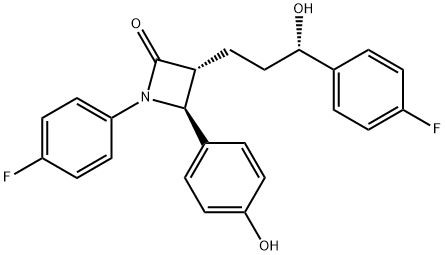
(3R,4S)-1-(4-Fluorophenyl)-3-[(S)-3-(4-fluorophenyl)-3-hydroxypropyl]-4-(4-hydroxyphenyl)azetidin-2-one;Ezetimibe
CAS NO.:163222-33-1
Empirical Formula: C24H21F2NO3
Molecular Weight: 409.43
MDL number: MFCD00937872
EINECS: 682-606-0
| Pack Size | Price | Stock | Quantity |
| 50MG | RMB26.40 | In Stock |
|
| 250MG | RMB74.40 | In Stock |
|
| 1g | RMB145.60 | In Stock |
|
| 5g | RMB472.80 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 164-166°C |
| alpha | D22 -33.9° (c = 3 in methanol) |
| Boiling point: | 654.9±55.0 °C(Predicted) |
| Density | 1.334±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Soluble in DMSO (up to 25 mg/ml) or in Ethanol (up to 15 mg/ml) |
| pka | 9.72±0.30(Predicted) |
| form | powder |
| color | White or off-white |
| BCS Class | 2 |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months. |
| InChI | InChI=1S/C24H21F2NO3/c25-17-5-1-15(2-6-17)22(29)14-13-21-23(16-3-11-20(28)12-4-16)27(24(21)30)19-9-7-18(26)8-10-19/h1-12,21-23,28-29H,13-14H2/t21-,22+,23-/m1/s1 |
| InChIKey | OLNTVTPDXPETLC-XPWALMASSA-N |
| SMILES | N1(C2=CC=C(F)C=C2)[C@H](C2=CC=C(O)C=C2)[C@@H](CC[C@@H](C2=CC=C(F)C=C2)O)C1=O |
| CAS DataBase Reference | 163222-33-1(CAS DataBase Reference) |
| EPA Substance Registry System | 2-Azetidinone, 1-(4-fluorophenyl)-3-[(3S)-3-(4-fluorophenyl)-3-hydroxypropyl]-4-(4-hydroxyphenyl)-, (3R,4S)- (163222-33-1) |
Description and Uses
Ezetimibe is a once-daily orally active cholesterol absorption inhibitor, launched as a hypolipidemic agent. The one-step diastereo- and enantioselective formation of β-lactams starting from commercially available (3S)-hydroxy-y-lactone is the key point of the asymmetric synthesis of ezetimibe. The 2-azetidinone class was initially designed as acylcoenzyme A: cholesterol acyltransferase (ACAT) inhibitors but experimental data suggest that this compound acts in the intestinal wall to inhibit cholesterol through a novel mechanism with an as yet undiscovered target. Orally administered ezetimibe inhibited increases in plasma cholesterol in four cholesterol-fed animals species (hamster, rats, dogs and rhesus monkeys). In rats cannulated in the intestine and bile duct, [3H]-ezetimibe inhibited cholesterol absorption by more than 95%. In cholesterol-fed LDL receptor+apoE knockout mice, treatment with ezetimibe reduced atherosclerotic lesion cross sectional area by 48% in the aorta and 20% in the carotid artery. Moreover, the plasma cholesterol levels were reduced and the progression of lesions was inhibited. Ezetimibe is highly protein bound and is metabolized by the liver to its glucuronide metabolite, which represents 80-90% of circulating ezetimibe. About 90% of ezetimibe and/or the glucuronide metabolite are excreted in the feces and 10% in the urine. The parent compound and its glucuronide metabolite undergo enterohepatic recirculation; in consequence, the drug is slowly eliminated. In hypercholesterolemic patients, ezetimibe (10 mglday, 12 weeks) reduced LDL cholesterol by 18% and total cholesterol by 12%, with a similar safety profile to placebo. Co-administration of ezetimibe with statins or fenofibrate lowered LDL cholesterol levels more than either monotherapy. Ezetimibe was well tolerated and interaction studies provided evidence that ezetimibe had no significant effect on the activity of major CYP450 drug-metabolizing enzymes. Moreover, no pharmacokinetic/pharmacodynamic interactions were seen between ezetimibe and statins and others frequently administered drugs. .
For use as adjunctive therapy to diet for the reduction of elevated total-C, LDL-C, and Apo B in patients with primary (heterozygous familial and non-familial) hypercholesterolemia.
Safety
| Symbol(GHS) |  GHS09 |
| Signal word | Warning |
| Hazard statements | H410 |
| Precautionary statements | P273-P391-P501 |
| Risk Statements | 36/37/38 |
| Safety Statements | 26-36-24/25 |
| HS Code | 29337900 |
| Hazardous Substances Data | 163222-33-1(Hazardous Substances Data) |