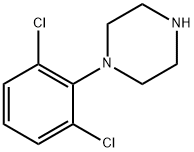
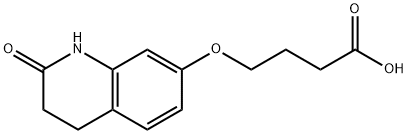
Aripiprazole , ≥99% , 129722-12-9
Synonym(s):
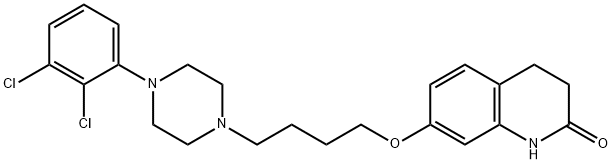
7-[4-[4-(2,3-Dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydro-2(1H)-quinolinone;7-{4-[4-(2,3-Dichlorophenyl)-1-piperazinyl]butoxy}-3,4-dihydro-2(1H)-quinolinone;Aripiprazole
CAS NO.:129722-12-9
Empirical Formula: C23H27Cl2N3O2
Molecular Weight: 448.39
MDL number: MFCD00892072
EINECS: 603-355-5
| Pack Size | Price | Stock | Quantity |
| 1G | RMB39.20 | In Stock |
|
| 5G | RMB104.80 | In Stock |
|
| 25G | RMB335.20 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 139°C |
| Boiling point: | 646.2±55.0 °C(Predicted) |
| Density | 1.263±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: soluble5mg/mL, clear (warmed) |
| pka | 14.42±0.20(Predicted) |
| form | powder |
| color | white to beige |
| Merck | 14,785 |
| BCS Class | 2 |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. |
| InChI | InChI=1S/C23H27Cl2N3O2/c24-19-4-3-5-21(23(19)25)28-13-11-27(12-14-28)10-1-2-15-30-18-8-6-17-7-9-22(29)26-20(17)16-18/h3-6,8,16H,1-2,7,9-15H2,(H,26,29) |
| InChIKey | CEUORZQYGODEFX-UHFFFAOYSA-N |
| SMILES | N1C2=C(C=CC(OCCCCN3CCN(C4=CC=CC(Cl)=C4Cl)CC3)=C2)CCC1=O |
| CAS DataBase Reference | 129722-12-9(CAS DataBase Reference) |
Description and Uses
Aripiprazole was launched for the treatment of psychoses including schizophrenia and offers a novel mechanism of action as a partial D2 receptor agonist. Aripiprazole can be synthesized in three steps beginning by the condensation of 7-hydroxy-1,2,3,4- tetrahydroquinolin-2-one with 1 ,Cdibromobutane followed by reaction with 1-(2,3- dichlorophenyl)piperazine. Aripiprazole is a significant D2 agonist/antagonist, 5-HT2 antagonist and 5-HT1α agonist combined with minimal affinity for a,-adrenergic, H1 and M1 receptors. It has a low D4:D2 selectivity ratio and a D2:5-HT2 affinity ratio that exceeds 15; resulting in different pharmacological characteristics compared to other atypical antipsychotics agents such as clozapine. In animal models, aripiprazole inhibits apomorphine-induced stereotypy without causing catalepsy and ptosis. Moreover, in contrast to classical antipsychotics that produce disabling movement disorders, aripiprazole does not cause an upregulation of D2 receptors or an increase in immediate early gene expression of e.g. the c-fos mRNA in the striatum. In patients with acute relapse of schizophrenia, treatment with aripiprazole provided significant improvement in both positive and negative syndrome scale (PANSS) total score in both short- and longterm evaluations. These results were comparable to those observed with haloperidol or risperidone; however, the early response rate was greater with aripiprazole. Aripiprazole was well tolerated with mild to moderate adverse events such as nausea, dizziness, somnolence and weight gain. The rates of extrapyramidal symptoms were lower than with haloperidol, prolactin levels increase has been uncommon and no significant Q-Tc interval prolongation was observed compared with placebo. Finally, studies suggested a minimal impact of aripiprazole administration on total cholesterol levels and on fasting blood sugar in contrast to other antipsychotics. Aripiprazole has a bioavailability of 87%, a tmax of 3-5 h and a half-life time of 48-68 h. Aripiprazole has been found to have linear kinetics and is mainly metabolized via the cytochrome systems CYP2D6 and CYP3A4. It has little effect on the blood levels of other medications; interaction with both lithium and divalproex sodium found minimal impact. Aripiprazole has also been studied in other psychiatric disorders, including bipolar disorders and has shown great efficacy.
For the treatment of schizophrenia and related psychotic disorders.
Safety
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225-H301+H311+H331-H370 |
| Precautionary statements | P210-P233-P280-P301+P310-P303+P361+P353-P304+P340+P311 |
| Hazard Codes | F,Xn |
| Risk Statements | 11-20/21/22-36 |
| Safety Statements | 16-36/37 |
| RIDADR | UN 1993C 3 / PGIII |
| WGK Germany | 3 |
| RTECS | VC8275950 |
| HS Code | 29349990 |
| Hazardous Substances Data | 129722-12-9(Hazardous Substances Data) |
| Toxicity | TDLo ivn-dog: 0.03 mg/kg/10M TXAPA9 173,120,2001 |