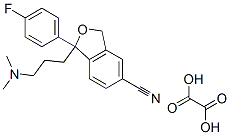
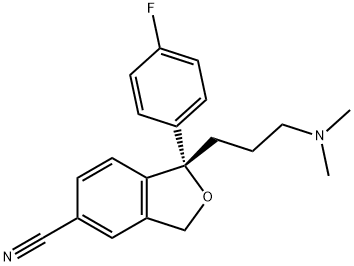
(S)-Citalopram-d6Oxalate
Synonym(s):
Escitalopram oxalate;S-(+)-1-[3-(Dimethylamino)propyl]-1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile oxalate;S-(+)-Citalopram oxalate
CAS NO.:
Empirical Formula: C22H23FN2O5
Molecular Weight: 414.43
MDL number: MFCD06407826
EINECS: 620-544-8
| Pack Size | Price | Stock | Quantity |
| 1mg | RMB2436.80 | In Stock |
|
| others | Enquire |
PRODUCT Properties
| Melting point: | 152-153°C |
| alpha | D +12.31° (c = 1 in methanol) |
| storage temp. | 2-8°C |
| solubility | DMSO: ≥15mg/mL |
| form | powder |
| color | white to tan |
| Merck | 14,2318 |
| BCS Class | 1 |
| Stability: | Hygroscopic |
| InChI | InChI=1S/C20H21FN2O.C2H2O4/c1-23(2)11-3-10-20(17-5-7-18(21)8-6-17)19-9-4-15(13-22)12-16(19)14-24-20;3-1(4)2(5)6/h4-9,12H,3,10-11,14H2,1-2H3;(H,3,4)(H,5,6) |
| InChIKey | KTGRHKOEFSJQNS-UHFFFAOYSA-N |
| SMILES | C1(CCCN(C)C)(C2=CC=C(F)C=C2)C2=C(C=C(C#N)C=C2)CO1.C(O)(=O)C(O)=O |
| CAS DataBase Reference | 219861-08-2(CAS DataBase Reference) |
Description and Uses
Escitalopram was launched as Cipralex? in Switzerland, Sweden and UK for the treatment of depression and panic disorder. It is the S-enantiomer version of the selective serotonin reuptake inhibitor (SSRI) citalopram approved in 1989. It can be obtained from 5cyanophthalide by successive reactions with 4-fluorophenyl magnesium bromide and 3-(dimethylamino)propyl magnesium chloride. The resulting racemic diol can be resolved by several routes such as crystallization with a chiral acid. Finally, a two step cyclisation procedure affords escitalopram. Escitalopram is twice as effective as the racemate and over 100 fold more potent than the R-enantiomer in inhibiting the 5HT reuptake in vivo in rat brain synaptosomes. Moreover, it exhibits higher selectivity for the human serotonin transporter relative to the noradrenaline or dopamine transporters than any other currently available SSRl’s. In the mouse forced swim test, the duration of immobility (which reflects antidepressant activity) for escitalopram was comparable to citalopram and greater than (R)-citalopram. Clinical trials in patients with panic disorders or depression have shown that escitalopram has a clinically relevant and significant effect. Additionally, it has a faster onset of antidepressant action than citalopram. Escitalopram has linear pharmacokinetics, with a long half-life (27-32 h). It is extensively metabolized in the liver via cytochromes P450 to S(+)-desmethyl and S(+)-didesmethyl citalopram. However, it has been shown to be a weak or negligible inhibitor of CYP450 drugmetabolizing enzymes in vitro. Escitalopram is well tolerated with nausea being the most common side effect.
A labelled inhibitor of serotonin (5-HT) uptake. Antidepressant
Safety
| Symbol(GHS) |  GHS07 |
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 |
| Hazard Codes | Xi |
| Risk Statements | 36/37/38 |
| Safety Statements | 26-36 |
| WGK Germany | 3 |
| RTECS | NP6333500 |
| HS Code | 29322090 |